Launch of the “U.S. Export Cosmetics Label Review” AI Service! Be Sure to Use It When Creating Packaging.
Hello, this is Packative. If you’re exporting cosmetics to the U.S. or preparing to do so, please pay close attention to this post.👀 Unlike Korea, cosmetics for the U.S. market must comply with FDA regulations for labels, ingredients, and warning statements. If even a small label does not meet the requirements, exports can be delayed...

Hello, this is Packative.
If you’re exporting cosmetics to the U.S. or preparing to do so, please pay close attention to this post.👀
Unlike Korea, cosmetics for the U.S. market must comply with FDA regulations for labels, ingredients, and warning statements. If even a small label does not meet the standards, exports may be delayed, so careful review from the packaging development stage is essential.
At Packative, we are making various AI-based efforts to support the digital transformation of the packaging industry. For brand managers, designers, and export preparation teams working on cosmetics for the U.S. market, we’ve launched an AI service called “U.S. Export Cosmetics Label Review” that automatically checks export regulations.🤖💄
When creating cosmetics packaging for the U.S. market, what should you watch out for? feat. required label information
When producing cosmetics packaging for U.S. export, you need to follow the FDA’s labeling requirements. According to FDA regulations, the following information must be shown in English on the cosmetics label.
1. Identity of the Product
- It must clearly indicate that the product is a cosmetic.
- Example: Moisturizing cream, Shampoo
2. Net Quantity of Contents
- The volume or weight must be stated.
- U.S. customary units should be used first, with metric units added if needed.
- Example: Net Wt. 4 oz(113g)
3. Ingredients
- Use INCI (International Nomenclature of Cosmetic Ingredients) names
- List in descending order of concentration: from highest to lowest
- Allergens may be highlighted separately if needed
4. Name and Place of Business
- The company name and address responsible for U.S. distribution are required
- Example: Distributed by ABC Inc, New York, NY 10001
5. Warnings/Cautions
- Include as needed depending on the product
- Example: For external use only, Avoid contact with eyes
Check the differences between Korean cosmetics labels and U.S. export labels in the table below.✅
| Item | Korea 🇰🇷 | U.S. 🇺🇸 |
|---|---|---|
| Label language | Korean required, foreign languages may be included | English required |
| Product name labeling | Functional claims may be indicated | Functional claims are generalized as 'cosmetic'; functional products are subject to separate FDA OTC regulations |
| Ingredient listing | INCI + Korean may be used together | INCI only allowed |
| Manufacturer/seller information | Manufacturer and responsible seller must be stated | U.S. importer/distributor information required |
| Expiration date / period after opening | 'Expiration date' or 'period after opening' required | Expiration date is recommended; period after opening may be shown (optional) |
| Functional cosmetics | Functional certification required | FDA-regulated functional products (e.g. sunscreens) are subject to separate OTC rules |
Packative AI tools - Label review, now done with AI in just minutes!
What are the benefits of the AI label review feature?
- Speed: What used to take hours to days can now be handled in just minutes with a single AI review.
- Accuracy: Reduce mistakes in advance based on complex FDA regulations.
- Usability: Translation suggestions and regulatory references are provided together, making it ready for immediate use in practice.


Packative AI tools - Label review AI, how do you use it? A very simple process
With this intuitive process, anyone can easily use it as long as they have a label image.
STEP1. File upload
Please upload a Korean package design, PDF, product photo, and more.
 Packative AI tools_U.S. Export Cosmetics Label Review AI service
Packative AI tools_U.S. Export Cosmetics Label Review AI service
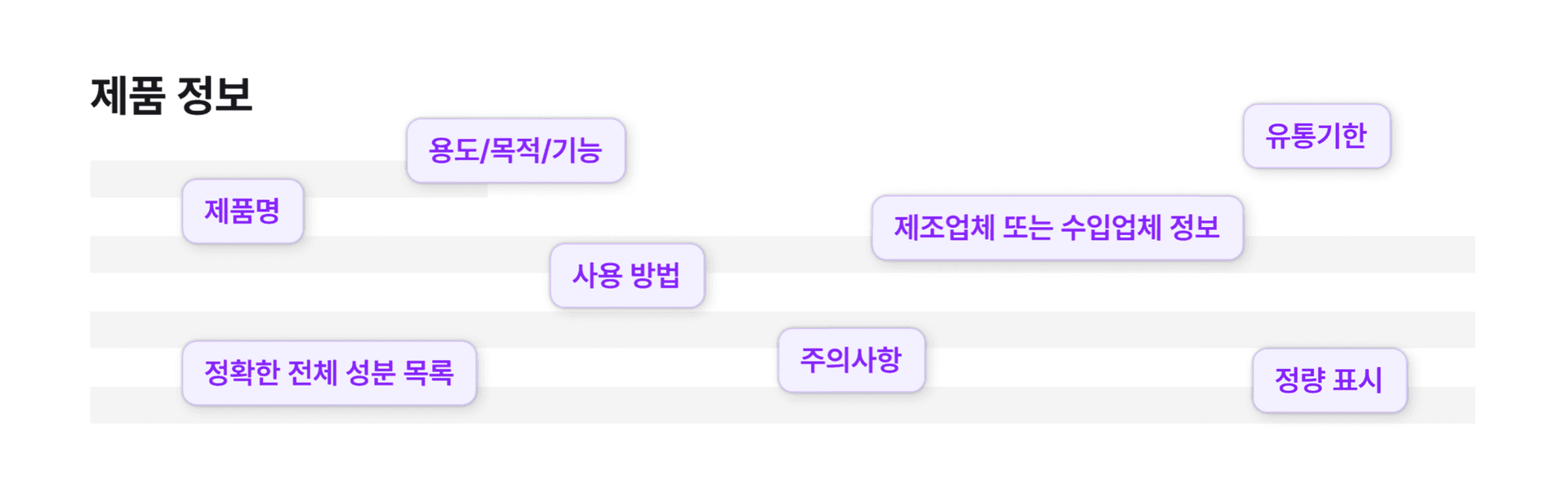
STEP2. Automatic extraction & analysis
The AI automatically recognizes key label elements such as ingredient names, directions for use, and cautions.
 Packative AI tools_U.S. Export Cosmetics Label Review AI service
Packative AI tools_U.S. Export Cosmetics Label Review AI service
STEP3. FDA compliance review
Detailed compliance is checked against the label regulations for cosmetics exported to the U.S.
 Packative AI tools_U.S. Export Cosmetics Label Review AI service
Packative AI tools_U.S. Export Cosmetics Label Review AI service
STEP4. Guidance on missing items
Any missing items or items that need additional review are shown along with the relevant FDA regulations.
 Packative AI tools_U.S. Export Cosmetics Label Review AI service
Packative AI tools_U.S. Export Cosmetics Label Review AI service
STEP5. Download report (coming soon)
The AI-reviewed results are being prepared so that you can download a report and apply it directly to your work.🏃🏻♀️➡️
 Packative AI Tools_U.S. Export Cosmetics Label Review AI service
Packative AI Tools_U.S. Export Cosmetics Label Review AI service
Packative AI tools - Label review AI, who is it for?
Packative’s AI tools can review U.S. export cosmetics labeling requirements in just a few minutes with a few clicks and even help fill in missing details. We recommend it for the following people.



- Brand managers: If you’re the brand manager responsible for this project, prepare U.S. export labeling with confidence.
- Package designers: For export cosmetics packaging design, don’t focus only on aesthetics—reflect regulatory compliance from the design stage.
- Export teams: Minimize delivery delays and risks caused by label errors.
Packative supports you in creating error-free packaging through automated AI review. If you’re curious about the U.S. Export Cosmetics Label Review AI service, click the image below to try it now.🚀
